The Chemical Basis for Life
Essential Questions:
When does covalent bonding happen? How does covalent bonding affect reactions ? What is the use of Atoms ? |
Objective:
- to describe the properties of water and how their functions
- explain the how enzymes act as catalyst in certain reactions
- describe temperature, pH, and concentration levels affect enzyme functions.
- describe the use of Atoms
- to describe the properties of water and how their functions
- explain the how enzymes act as catalyst in certain reactions
- describe temperature, pH, and concentration levels affect enzyme functions.
- describe the use of Atoms
.Key Terms:
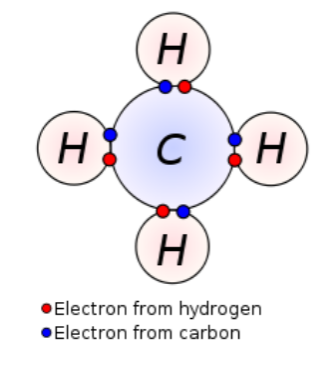
Covalent bonding- the sharing of electron pairs between atoms.
Polar molecules - the ends of a molecule called dipoles carry an unequal charge
Hydrogen bond - attraction between polar molecules in which hydrogen is bound to a larger atom
Nonpolar molecules - electrons equally in a covalent bond there is no net electrical charge across the molecule
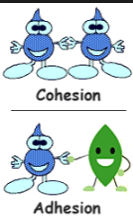
Adhesion - the force of attraction between different substances, such as glass and water
Cohesion - the force of attraction between molecules of the same substance
DNA- the material that make up genes and chromosomes stretched and looks like a double RNA
Proteins - sequences of amino acids, structural, hormonal, transportation functions in the body.
Covalent bonding- the sharing of electron pairs between atoms.
Polar molecules - the ends of a molecule called dipoles carry an unequal charge
Hydrogen bond - attraction between polar molecules in which hydrogen is bound to a larger atom
Nonpolar molecules - electrons equally in a covalent bond there is no net electrical charge across the molecule
Adhesion - the force of attraction between different substances, such as glass and water
Cohesion - the force of attraction between molecules of the same substance
DNA- the material that make up genes and chromosomes stretched and looks like a double RNA
Proteins - sequences of amino acids, structural, hormonal, transportation functions in the body.

Summary:
Within the basis for life are put together they have a lot of things to take in, atoms are the introduction of all the things after. Atoms have three separate pieces that they are made of protons, neutrons, and electrons. The protons have a positive charge, neutrons have no charge. Electrons have an orbit around the nucleus, they have a negative charge. Through the chemical basis for life, there are chemical reactions, in the process of chemical reactions the atoms made. Just as much as chemical reactions there are water properties, the water properties are what makes of a molecular structure. Water keeps the molecular structure because it consist of two hydrogen atoms and one oxygen atom, the atoms are held together by covalent bonds. In covalent bonding atoms do not share an equal amount of electrons at all times, if there is an equal amount of electrons being shared that will make it become non polar, if there is an unequal amount of electrons being shared than it will become polar. Water is a polar molecule, it can break down molecules that are also polar, non polar and polar reactions come from adhesion and cohesion. Adhesion is how water sticks to other substances, it is hydrogen bonding interactions with water and molecules that do not consume water. Cohesion allows the water molecule to stick to itself by attracting other water molecules than stick to another thing, like a penny. Within our bodies, there are pH scales, they determine how acidic or base solutions can function in your body.
Within the basis for life are put together they have a lot of things to take in, atoms are the introduction of all the things after. Atoms have three separate pieces that they are made of protons, neutrons, and electrons. The protons have a positive charge, neutrons have no charge. Electrons have an orbit around the nucleus, they have a negative charge. Through the chemical basis for life, there are chemical reactions, in the process of chemical reactions the atoms made. Just as much as chemical reactions there are water properties, the water properties are what makes of a molecular structure. Water keeps the molecular structure because it consist of two hydrogen atoms and one oxygen atom, the atoms are held together by covalent bonds. In covalent bonding atoms do not share an equal amount of electrons at all times, if there is an equal amount of electrons being shared that will make it become non polar, if there is an unequal amount of electrons being shared than it will become polar. Water is a polar molecule, it can break down molecules that are also polar, non polar and polar reactions come from adhesion and cohesion. Adhesion is how water sticks to other substances, it is hydrogen bonding interactions with water and molecules that do not consume water. Cohesion allows the water molecule to stick to itself by attracting other water molecules than stick to another thing, like a penny. Within our bodies, there are pH scales, they determine how acidic or base solutions can function in your body.